The World of Osmometry
Explore our ProductsAbout Osmometry
Osmometers are analytical instruments used to determine the osmotic concentration of solutions, primarily in aqueous solutions. They have diverse applications across various fields, including clinical laboratories for blood and urine tests, the biopharmaceutical industry for drug development, and the food and chemical industries for quality control and research.
An osmometer is a device used in Hospitals, clinical laboratories and diagnostic centres and research industry for measuring the concentration of particles in a solution, known as the osmolar concentration. This quantity can be expressed as osmolality (in mmol/kg) or osmolarity (in mmol/L). Clinical laboratories typically measure osmolality, which is considered more precise as weight is temperature independent. In laboratory analysis, a dissolved substance is referred to as a solute, and the substance in which the solute is dissolved is referred to as a solvent. A solute dissolved into a solvent creates a solution.
The unit for osmolar concentration or osmolality is milliosmole (abbreviated mOsm or mOsmol). For nonelectrolytes, 1 mmol equals 1 mOsm, whereas for electrolytes, the number of particles in a solution depends on the electrolyte's dissociation. When a solute is dissolved into a solvent, 4 colligative properties of the solvent change.
These properties include:
- Osmotic pressure
- Vapor pressure
- Boiling point
- Freezing point
Measurement Principles
Osmometry is an analytical measuring method for determining the osmotic value / pressure (often referred to as simply the osmolality) of a sample. The osmolality is defined as the concentration of all dissolved – and thus osmotically effective – particles in a solution based on 1 kilogram of solvent. The unit of osmolality is Osm/kg or Osmol/kg. The freezing point of a sample changes depending on the concentration of dissolved substances. Pure water has a freezing point of 0°C. The solution of one or several substances in water leads to lowering of the freezing point. A solution with an osmotic value of 1 Osmol/kg has a freezing point of -1.858°C. Through the linear correlation between the freezing point of a sample and its osmolality, freezing point osmometry provides high-precision analytics.
Measurement Method - Technology
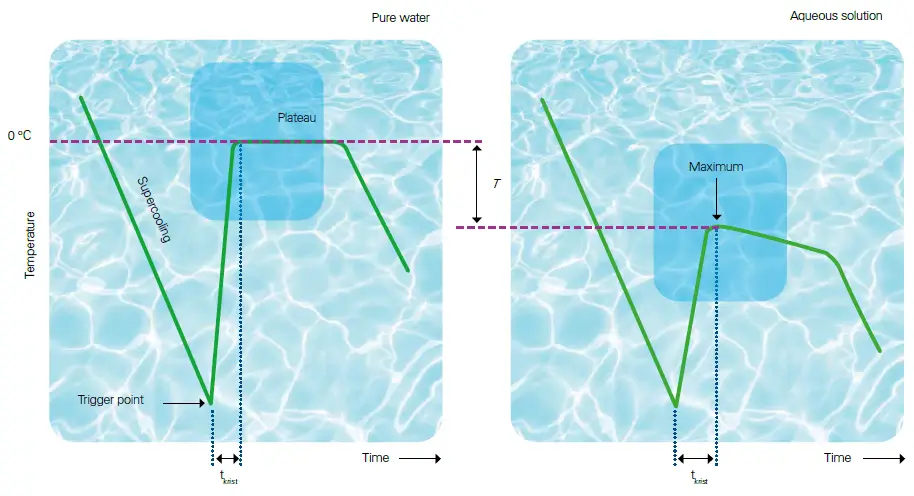
At the beginning of the measurement, the sample is supercooled in the lower cooling system. The freezing process of the sample material is released in a controlled manner by injecting an ice crystal using the release/triggering needle.The crystallization process leads to the release of thermal energy and the temperature of the sample increases until a plateau phase or a maximum is reached, which depicts the actual freezing point of the sample. At this point, the temperature is measured with an accuracy of 0.001°C using a high-precision temperature sensor.
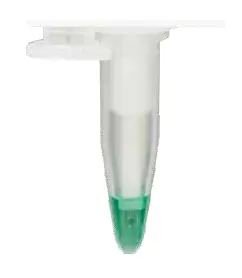
Position measuring vessel on the thermistor probe.
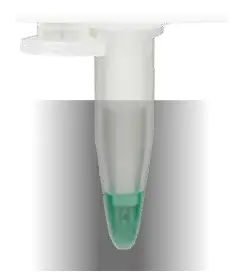
Sample is moved into the lower cooling system and cooled to less than 0°C by a microprocessor-controlled peltier element.
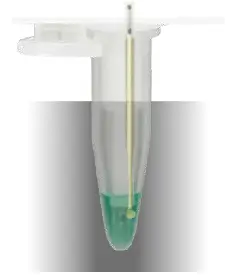
Injection of an ice crystal by cooled triggering needle (cryst-needle).
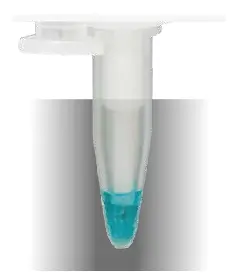
Crystallization of the sample material.
Measurement Principles - Physics
The freezing point is determined by super-cooling the solution several degrees below the solvent's freezing point and controlled initiation of the crystallization of the sample. The osmolality is computed from the freezing point depression. Super-cooling of the solution below freezing point. Due to the rapid cooling the sample does not freeze immediately. Crystallization is induced by injection of ice crystals and release of crystallization heat. During crystallization of the sample the heat of formation is released which increases the temperature of the solution until the freezing point is reached (plateau/maximum). The presence of solute particles predictably lowers the freezing point of the solvent. The plateau or slight slope of the temperature curve persists until complete crystallization, followed by a sharp drop in the curve upon complete crystallization.
Areas of Application
The FreezePoint® and Vapro® osmometers can complement each other and may overlap in certain applications. They should be promoted particularly in scenarios where one is more suited than the other. Below are the most commonly recommended applications for each osmometer, though their use is not restricted to these areas.



Applications
- General medicine
- Sports laboratories
- Forensic medicine
- Electron microscopy
- Physiology
- Clinical laboratories
- Botany
- Pediatrics
- Gynecology
- Military hospitals
- In-vitro-fertilization
- Urology
- Nephrology
- Hemodialysis
- Veterinary medicine
- Intensive care laboratories
- Pharmacy
- Dispensaries
- Contact lens manufacturing
- Food/beverage manufacturing
VQC Program
Online, real-time quality control evaluation and comparison program. Assists with evaluating the performance of your osmometer by comparing your QC results to those of other laboratories worldwide using the same instrument and lot of controls. Monthly reports assist with routine record-keeping Participation in a peer group comparison program fulfills good lab practice guidelines. Provided at no additional charge.
Peer Reviews from Select Science
"Reliable, accurate, and easy-to-use instrument for measuring osmolality."
The FreezePoint® Freezing Point Osmometer is a highly reliable and accurate instrument designed for measuring osmolality in various applications.
It offers easy operation and calibration, requires minimal maintenance, and delivers precise results with a measurement range of 0-2000 mOsm/kg and an accuracy of ± 2 mOsm/kg.
Its speed is also notable, providing results in as little as 60 seconds. Overall, it's an excellent choice for medical, pharmaceutical, and industrial laboratories.
"Beautiful equipment, essentiel for any lab that need to measure precise osmolarity."
Very easy to set up calibrate and use. I had an old Osmomat bought in the years which when I switched it on after more than a decade in storage worked perfectly first time and didn't even need to be recalibrated (which we did anyway but it was perfect). We upgraded last year and bought the osmomat 300 which use the same principle but can be plugged and monitor by a computer and send results on our network. It works perfectly, very reliable and very consistent results. It is sensitive and of course the imprecision increases with the concentration of the solutions but even for multimoles/kg solutions it is under 1%.
I highly recommend this product to anyone who need to check their solutions osmolarity. We work with expensive cell lines, primary cells, plasma samples, extracellular fluids, different medium etc. our results depend on precise osmolarity and it saved us massive headaches and lot of money to be able to set it precisely and to check the osmolarity of the mediums we bought.
"The reliability, accuracy and precision of the unit is what separates it from other units."
I purchased a vapor pressure osmometer back in 2017 when I established a stability lab to test cell culture media, process buffers and buffers both internal and external customers.
The unit has been very reliable, precise, accurate and easy to use.
I have most recently had more than five chemists in my lab using the unit. Its flexibility to interphase with LIMS has greatly improved GMP compliance and the quality of the data we generate.
This product has more potential in our current science spheres and could be enhance with more friendly user interphase.
Advantages
- Robust, precise, reliable, fast
- Small sample volume
- Easy control via the integrated touch screen display
- Step-by-step guidance through all measuring functions and setting options
- 2 or 3 point calibration
- Automatic and safe calibration with the Bruker Calibration Standards
- Data transfer to PC or laptop via RS-232 or USB
- Quality made in Germany
- Over 40 years of experience in precision measurement technology and osmometry
- Comprehensive advice and reliable service from our expert
- Eight user-selectable languages